Why Did They Do It That Way? Prescription Drugs
Does Medicaid cover prescription drugs? Under federal law, coverage of prescription drugs through the Medicaid program is an optional benefit rather than a mandatory one. However, all Medicaid agencies – from the states, DC, and the five US Territories – have elected to cover prescription drugs through their programs.
Prescription drugs are a core component of the United States’ health care system, with about half of all Americans using a prescription drug in the last 30 days. Although pharmaceutical innovations have driven improvements in length and quality of life, the growth in the cost of prescription drugs presents challenges to payers, including Medicaid. How does Medicaid provide access to prescription drugs, and what strategies are Medicaid agencies using to manage costs?
Does Medicaid cover prescription drugs?
Under federal law, coverage of prescription drugs through the Medicaid program is an optional benefit rather than a mandatory one. However, all Medicaid agencies – from the states, DC, and the five US Territories – have elected to cover prescription drugs through their programs. Unlike Medicare and private health insurance enrollees, Medicaid members do not pay significant costs for prescription drugs; although Medicaid agencies can elect to charge co-payments for prescriptions, these co-pays are typically not more than four dollars. Additionally, as stipulated in the Medicaid Drug Rebate Program (MDRP), Medicaid agencies cannot outright deny coverage of most drugs.

Medicaid provides access to health care for a large proportion of the population – about 78.5 million people in 2025 – resulting in a high volume of prescription drug claims. In FY2023, Although prescription drug spending is low in Medicaid compared to other payers, like private insurance and Medicare, prescription drug costs have grown substantially over the past several years, increasing 72% from FY2017 to FY2023. Much of this cost growth has been attributed to new, high-cost specialty drugs entering the market.
How do Medicaid agencies pay for prescription drugs?
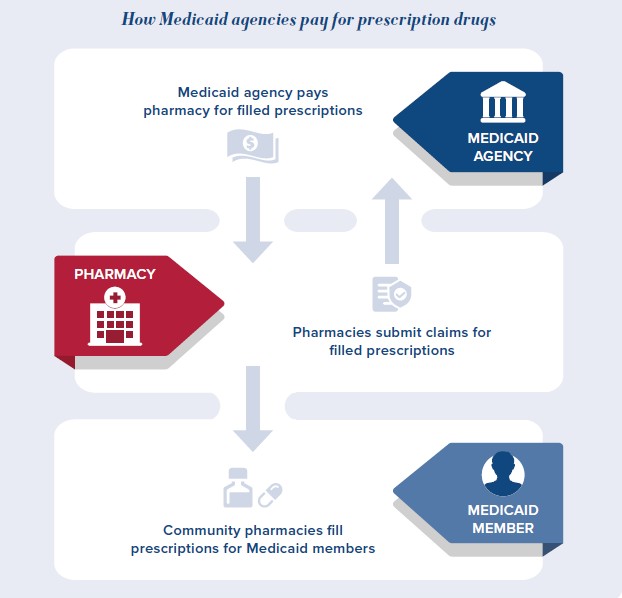
Like individuals who are covered by private insurance and Medicare, Medicaid members typically access prescription drugs through pharmacies in their communities. Depending on the state’s delivery system, the Medicaid agency or a Medicaid managed care organization then reimburses pharmacies for covered prescriptions that are filled by Medicaid members.
How much Medicaid agencies or managed care organizations pay pharmacies for these prescriptions is impacted by a range of regulatory requirements and market factors.
- In a fee-for-service delivery system, pharmacies are reimbursed based on two primary factors.
- 1) The drug’s “actual acquisition cost,” or the amount the pharmacy paid a drug manufacturer or wholesaler for the medication.
- 2) The dispensing fee covers costs associated with filling the prescription, such as pharmacists’ services and overhead costs.
- For drugs that are available from multiple manufacturers (such as drugs with both brand name and generic versions), state and federal regulations set maximum prices that the Medicaid agency can pay for the drug.
- In a managed care delivery system, there is more flexibility. Managed care organizations set their own pharmacy reimbursement rates, although federal regulations state that these rates must be sufficient to ensure Medicaid member access.
- Across both delivery systems, Medicaid agencies and managed organizations may contract with pharmacy benefit managers to handle certain administrative tasks, negotiate prices with manufacturers, and develop lists of “preferred drugs.”
Medicaid members may also receive prescription drugs as part of an inpatient hospital stay or receive drugs like immunotherapies or chemotherapies that are administered by a physician in an outpatient setting. Medicaid agencies reimburse providers for these drugs using separate formulas and reimbursement pathways.
What is the Medicaid Drug Rebate Program?
The Medicaid Drug Rebate Program (MDRP) was established in 1990 under the Omnibus Budget Reconciliation Act to help control Medicaid’s outpatient prescription drug costs. Under this program, drug manufacturers may enter a “national rebate agreement” with the Secretary of Health and Human Services (HHS). Through these agreements, drug manufacturers provide rebates to offset the cost of drugs covered by the Medicaid program. In return for these rebates, states must cover nearly all FDA-approved outpatient drugs from participating manufacturers, though they may impose preferred drug lists (PDLs), prior authorization requirements, or quantity limits to manage costs and ensure appropriate use.
The structure of the MDRP ensures that Medicaid consistently pays lower prices for prescription drugs than other payers. Rebates are calculated using a formula defined in statute, which considers whether a drug is brand-name or generic, its price in the broader market, and whether its price is increasing faster than inflation. To calculate these rebates, manufacturers participating in the program must report their data to CMS. AMP represents the average price wholesalers and retail pharmacies pay manufacturers for a given drug, excluding discounts or rebates. Best price refers to the lowest price available to any private purchaser, (excluding certain government programs such as the Veterans Health Administration). In effect, these rebates ensure that Medicaid programs pay at least as low as, or lower than, the lowest price available in the private market.
Over time, Congress has amended the formula used to calculate Medicaid rebates to offset rising prescription drug costs. Notably, the Affordable Care Act increased rebate amounts and extended rebates to Medicaid managed care organizations; previously, rebates had applied only to fee-for-service Medicaid. Under current law:
- For brand-name drugs, the rebate is the greater of 23.1% of AMP or AMP minus the best price, plus an additional rebate if the drug’s price increases faster than inflation.
- For generic drugs, the rebate is 13% of AMP, plus an additional rebate if the drug’s price increases faster than inflation

By requiring manufacturers to provide deep discounts while ensuring Medicaid members have access to nearly all FDA-approved drugs, the MDRP plays a critical role in containing costs within the Medicaid program. Medicaid rebates reduce drug spending by over 50%, with even larger reductions in fee-for-service Medicaid programs. In FY 2023, for example, Medicaid’s total gross spending on outpatient drugs was approximately $100 billion, but after rebates, net spending was $51 billion. The MDRP also helps contain prescription drug spending in comparison to other payers, with Medicaid typically receiving the lowest prices on drugs.
Even with these rebates, Medicaid agencies continue to report substantial concerns over the rising cost of prescription drugs. The MDRP has historically been effective at controlling pharmacy costs, but the proliferation of new, high-cost specialty drugs is placing increased pressure on state budgets. Because rebates are tied to average market price and inflation, new drugs with high prices can be very expensive for Medicaid agencies, even after rebates. With many new specialty drugs in the development pipeline, Medicaid agencies will need new tools to control the cost of prescription drugs, such as enhanced rebates.
How do Medicaid agencies ensure prescribed drugs are safe, effective, and cost efficient?
While the MDRP is an important tool for managing prescription drug spending, Medicaid agencies use additional tools to control costs, ensure safe prescribing of medications, and reduce misuse of prescription drugs.
Medicaid agencies use utilization management tools and review prescription drug claims to ensure the safe and efficient use of medications. Common strategies include:


What factors may impact Medicaid prescription drug coverage in the future?
Due to the MDRP, Medicaid typically pays less for prescription drugs than other payers. However, prescription drug spending has still grown substantially in Medicaid, with spending after rebates increasing from $30 billion in FY 2017 to $51 billion in FY 2023. Much of this cost growth has been attributed to new high-cost drugs, including cell and gene therapies, and new blockbuster drugs, such as glucagon-like peptide-1 (GLP-1) drugs.
High-cost specialty drugs. Over the past several years, innovative new treatments for complex, chronic, and rare diseases have been granted Food and Drug Administration (FDA) approval. Many of these drugs are cell and gene therapies, which use live cells or alter genes to treat conditions like cancer, sickle cell disease, and spinal muscular atrophy. These therapies have the potential to drive significant improvements in patient outcomes but often have high price points; many new cell and gene therapies cost over $1 million per dose and can have launch prices as high as $4 million dollars per dose. As more of these medications enter the market, they have the potential to drive spikes in Medicaid spending and create budget volatility for states. The long-term value of these drugs in terms of lifelong health improvements for patients may not necessarily result in budget savings for the Medicaid programs that initially cover their costs, as well, which poses broader questions about assessing value across payers in the health care system.
Blockbuster drugs. Blockbuster drugs are typically defined as pharmaceutical products with over $1 billion in annual sales. Although blockbuster drugs typically have lower launch prices than high-cost specialty drugs, high levels of utilization can lead to significant cost pressures on Medicaid programs. GLP-1 medications are a notable example. Medicaid agencies’ spending on these products almost doubled from 2022 to 2023 as utilization skyrocketed, even though Medicaid is only required to cover these drugs for non-weight loss indications such as diabetes. Similarly, utilization of certain cancer immunotherapies has increased dramatically over the past decade in Medicaid.
Conclusion
Pharmaceutical innovations can transform care for patients. Drugs in the development pipeline may offer new treatment options for debilitating conditions like cancer, hemophilia, muscular dystrophy, Alzheimer’s disease, and cardiovascular disease. Medicaid plays an essential role in helping people access these medications, but as new high-cost and blockbuster drugs come to market, prescription drug spending will increasingly strain Medicaid budgets. To ensure that Medicaid members have access to pharmaceutical innovations, while also maintaining the stability of state and territory budgets, Congress may need to explore new policy options like enhancing rebates through the Medicaid Drug Rebate Program.
Related resources
NAMD Comments on Proposed Rule that Would Require Medicaid Coverage of Anti-Obesity Medications
Breaking the Bottleneck: Modernizing Medicaid Drug Access in the Million Dollar Therapy Era
Stay Informed
Drop us your email and we’ll keep you up-to-date on Medicaid issues.